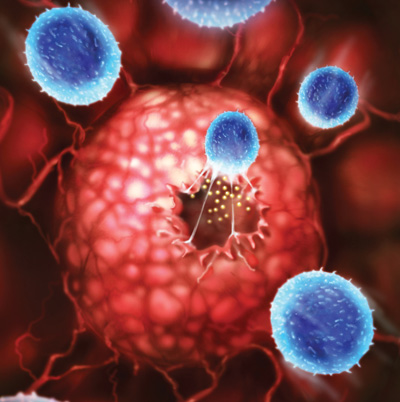
Cancer researcher Patrick Hwu, M.D., likes to think of the body’s immune system as the armed forces in charge of homeland defense and of tumor cells as homegrown terrorists. When everything is going well, the immune system recognizes malignant cells and destroys them. But like a general whose troops are not quite the crack soldiers he expected, Dr. Hwu has to admit, the terrorists too often get the better of his wimpy forces, which is why 560,000 Americans will die of cancer in 2009, the American Cancer Society forecasts. “We don’t really know why the immune system doesn’t keep most cancers in check,” says Dr. Hwu, a cancer immunologist at the M.D. Anderson Cancer Center in Houston. “The tumor might disguise itself from the immune system or make things that suppress the immune system.”
When he was working at the National Cancer Institute (NCI) in the 1990s, Dr. Hwu and colleagues decided they needed an innovative way to retrain the immune system. What if they could inject into cancer patients the very “markers” that stud the surface of malignant cells? That’s how vaccines for infectious diseases such as measles and smallpox work, by training the immune system to recognize and attack the markers on disease-causing viruses. In this case, Dr. Hwu and others reasoned that injecting “tumor antigens” might train the immune system to recognize the tumor cells as foreign and destroy them. It would be a “cancer vaccine” — not a vaccine like that for measles, which prevents a disease, but one that treats it. (These cutting-edge cancer vaccines are not to be confused with Gardasil, a vaccine that primes the immune system to attack the virus that causes cervical cancer. Very few human cancers are known to be caused by viruses, so “cancer vaccine” has come to mean one that treats existing cancer.)
The NCI scientists were hardly alone in dreaming of a cancer vaccine. The appeal was obvious: Oncologists’ cancer-fighting arsenal consisted of surgery, radiation, and chemotherapy — or as critics like to say, cutting, burning, and poisoning. Not only did the three weapons come with serious side effects, but they were far from universally effective. Perhaps harnessing the immune system would vanquish tumor cells with little to no collateral damage to the rest of the body. The idea took hold in a serious way in the 1970s, recalls Dr. Len Lichtenfeld of the American Cancer Society, “and it was going to be a slam dunk. We were going to cure cancer.” Obviously, that did not happen. Instead, vaccine after vaccine failed. But with advances in their understanding of both cancer and immunology, scientists finally scored some notable successes: Three large-scale studies, called Phase III clinical trials, presented at the 2009 meeting of the American Society of Clinical Oncology (ASCO), suggest that cancer vaccines may one day play a role in cancer treatment.

In one study, a vaccine based on the work of Dr. Hwu and his NCI colleagues extended what’s called progression-free survival — how long patients live before a cancer that has been treated with chemotherapy or radiation returns — in people with advanced melanoma, one of the most lethal cancers. The vaccine consists of antigens that stud melanoma cells. By injecting these “gp100” molecules into patients, “the vaccine activates the T cells of the immune system to recognize the antigens on the surface of the tumor,” says Dr. Hwu. “The T cells then secrete enzymes that poke holes in the tumor cells’ membrane, causing it to disintegrate” and the cancer cells to die.
This was the first time a melanoma vaccine had shown any benefit in a Phase III trial, but even with this distinction, it was no “slam dunk.” The 93 patients who received a standard therapy called interleukin-2 and no vaccine remained in remission for an average of 1.6 months. The 86 patients who received the vaccine plus interleukin-2 remained in remission for just under three months; hardly a big improvement. And on what every cancer patient cares about most — staying alive — the vaccine fell short. Vaccinated patients lived 17.6 months, on average, while unvaccinated patients lived 12.8 months, a difference that statistical analysis determined could be due to chance.
Why wasn’t the benefit greater? It’s possible that the patients did not have enough T cells to mount an effective immune response, says Dr. Hwu. Or maybe, for some reason, the T cells do not find the melanoma cells. Perhaps the tumor disguised itself, hiding the antigen that the T cells were trained to look for. “It’s possible that we did not have enough soldiers, or that they were not well-enough trained, or that they were not able to find the battlefield,” says Dr. Hwu. It’s also possible that a vaccine needs to contain more than the gp100 antigen. “Unique antigens specific to a patient’s cancer do exist, and using them might stimulate a more effective immune response,” he says. “But it would take months to synthesize such a custom-made vaccine,” and advanced melanoma patients do not have months.
Another study unveiled at ASCO this year, however, took the customization approach. Invented by M.D. Anderson’s Larry Kwak when he, too, was at NCI, the vaccine contains markers from each patient’s cancer — in this case, a form of non-Hodgkin’s lymphoma called follicular lymphoma, which is a cancer of (ironically) immune-system cells called B cells. After a biopsy harvests a patient’s malignant B cells from the lymph nodes, it takes about one month to make the vaccine. “The vaccine uniquely recruits the patient’s immune system to seek and destroy only tumor B cells,” said Stephen Schuster of the University of Pennsylvania, who led the study.
“It’s the ultimate in personalized therapy,” adds Dr. Kwak. “Even if patients have the same type of lymphoma, the tumors will still have different antigens, and that’s what you want your vaccine to contain.” The 76 patients who received the custom-made vaccine after a standard course of chemo remained disease-free just over one year longer than the 41 patients who received the chemo alone (44.2 months compared to 30.6 months). In an earlier Phase II study at NCI, patients have remained disease-free for an average of eight years.
Crucially, all the patients in the latest study were in complete remission or had no detectable cancer when they received their personalized vaccine. That is, they had responded to standard chemo and sustained a remission for six months. To skeptics, that raises the possibility that these patients’ cancer might have been less aggressive, and that fact — not the vaccine — accounts for their longer survival. In addition, the vaccine was compared to chemo that has now been superseded by something more effective. Oncologist Ron Levy of Stanford University, a founding father of cancer vaccines, gives BioVax ID “a qualified yes” in terms of effectiveness, but only a “maybe” on whether it will find a meaningful role in treating patients with follicular lymphoma.

Dr. Kwak sees it differently. He admits that the vaccine — and possibly all cancer vaccines — work best in patients whose tumor has shrunk to almost nothing or who are in complete remission. “Then you bring in the vaccine to mop up any remaining cancer cells,” he says. “That’s the strategy, and it should work for other cancers.” If he’s right, then the failures of other cancer vaccines might be because the patients in those studies had active and sometimes widespread cancer. “We now suspect that cancer vaccines will not work very well against advanced tumors,” says Dr. Kwak. “Tumors have the ability to turn off the immune response, and the more tumor you have, the more factors that can do that.”
That may explain why a vaccine for prostate cancer has fallen short. Called Provenge, it failed to beat back disease, let alone improve survival, in early tests, and the Food and Drug Administration refused to approve it in 2007. But the manufacturer, Dendreon, has spent an estimated $560 million on it and isn’t giving up. In a new trial, also presented at ASCO this year, the company announced that among men whose prostate cancer was no longer responding to hormone treatment or chemo, those who received Provenge lived four months longer than those who did not, 26 months versus 22 months. Just under one-third of the Provenge men were still alive after three years, compared to just under one-quarter of the men who did not receive the vaccine — 31 percent versus 23 percent. That’s far from a cure, but Dendreon is hopeful that it will get a green light from the FDA in 2010.
But don’t expect that to open the floodgates for cancer vaccines. So many have failed that it seemed as if they were going to be yet another therapy that looked terrific on paper, and even in lab animals, but which crashed and burned when tested in people. For instance, in 2006 a pancreatic-cancer vaccine called PANVAC-VF failed to improve overall survival. Considering that it was being compared to chemotherapy that keeps patients alive for a median three months, the result was a big disappointment.
The encouraging results for the melanoma and follicular lymphoma vaccines — and, to a lesser extent, for Provenge — have definitely been heard, breathing new life into a field that had been hanging by a thread. “Looking to the future, I think we’ll see a role for vaccines where the cancer is found very early, when the disease burden is small [because the patient is in remission], or when there are circulating cancer cells that can’t be eliminated any other way,” says Dr. Lichtenfeld. “But it looks unlikely that cancer vaccines will produce a response in people who have cancer throughout their system.” The latter, of course, are the sickest of the sick, so there is no small irony in the likelihood that this most innovative of cancer treatments will help not those who need it most, but those with an already hopeful prognosis.
Become a Saturday Evening Post member and enjoy unlimited access. Subscribe now


Comments
Cancer was cured in the 70’s and when the cured patient”- Steve McQueen”- was going to tell the world about the Dr. who found the cure, McQueen was suicided, whacked ,secretly stuck with an OD.,.
In big business, as in the working gals motto,- states”No money_No Honey”.
Mac